Healthcare
RESP® Biosensor
Manage lung health from hospital to home with objective, real-time monitoring of symptoms
FDA 510(k) Class II Device | CE Mark | HIPAA Compliant

Technology Overview
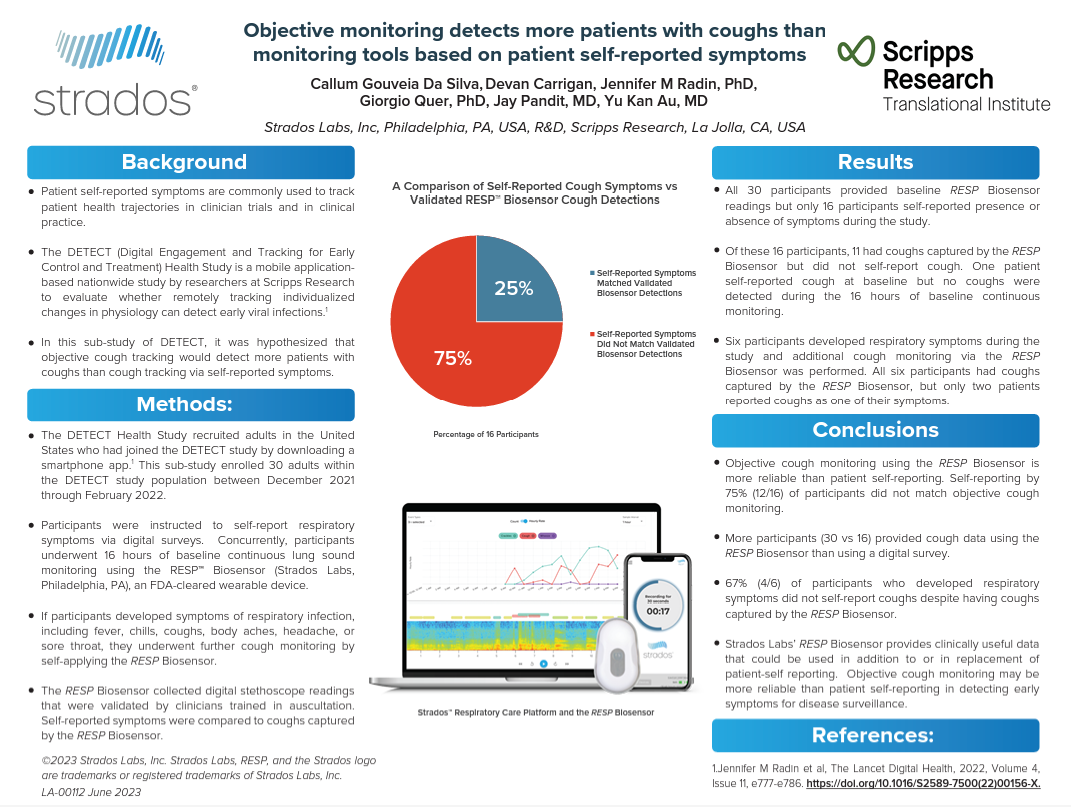
- The RESP® Biosensor offers clinicians an objective approach to monitoring early warning symptoms of exacerbations such as coughing and wheezing in high-risk patients
- Clinicians can view cough and wheeze data from patients in real-time to better understand respiratory status
- Patient and clinician-friendly, the RESP Biosensor reduces the burden on patients while improving clinician effectiveness
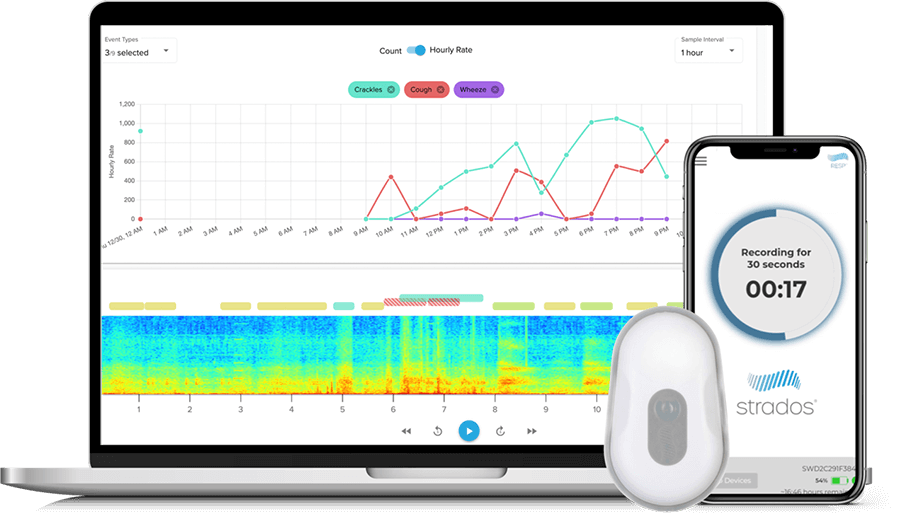
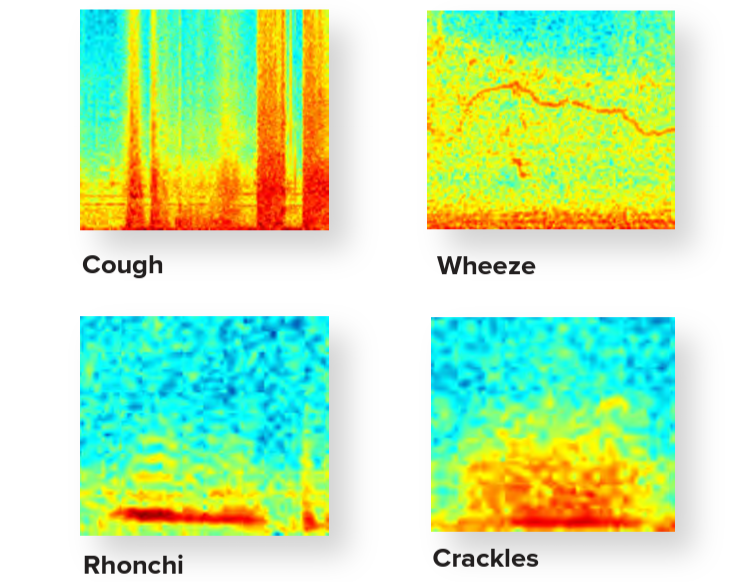
RESP® Biosensor Measurements
Identify changes in early warning signs of a COPD or Asthma flareup
Cough
Wheeze, Rhonchi & Crackles
Why Monitor Cough and Lung Sounds?
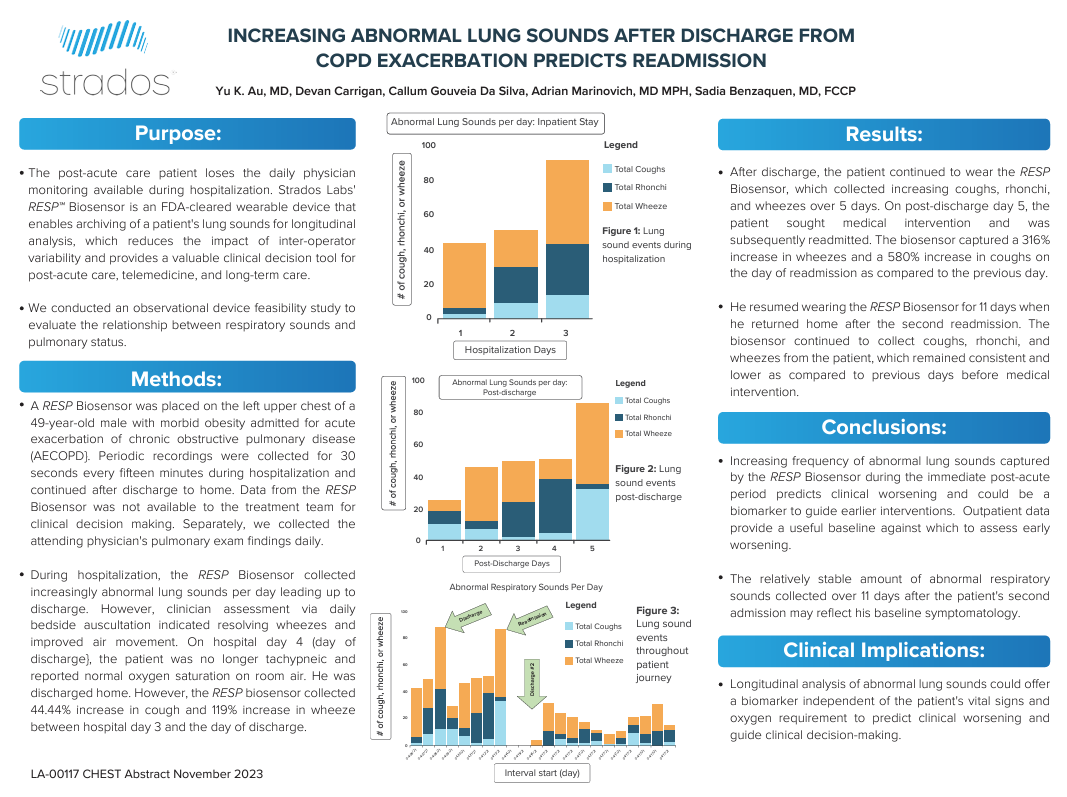
- 1 in every 5 patients discharged for COPD are readmitted within 30 days (source)
- Increases in cough have been shown to predict exacerbations (source)
- Studies show patients have difficulty tracking early warning signs such as cough and wheezing
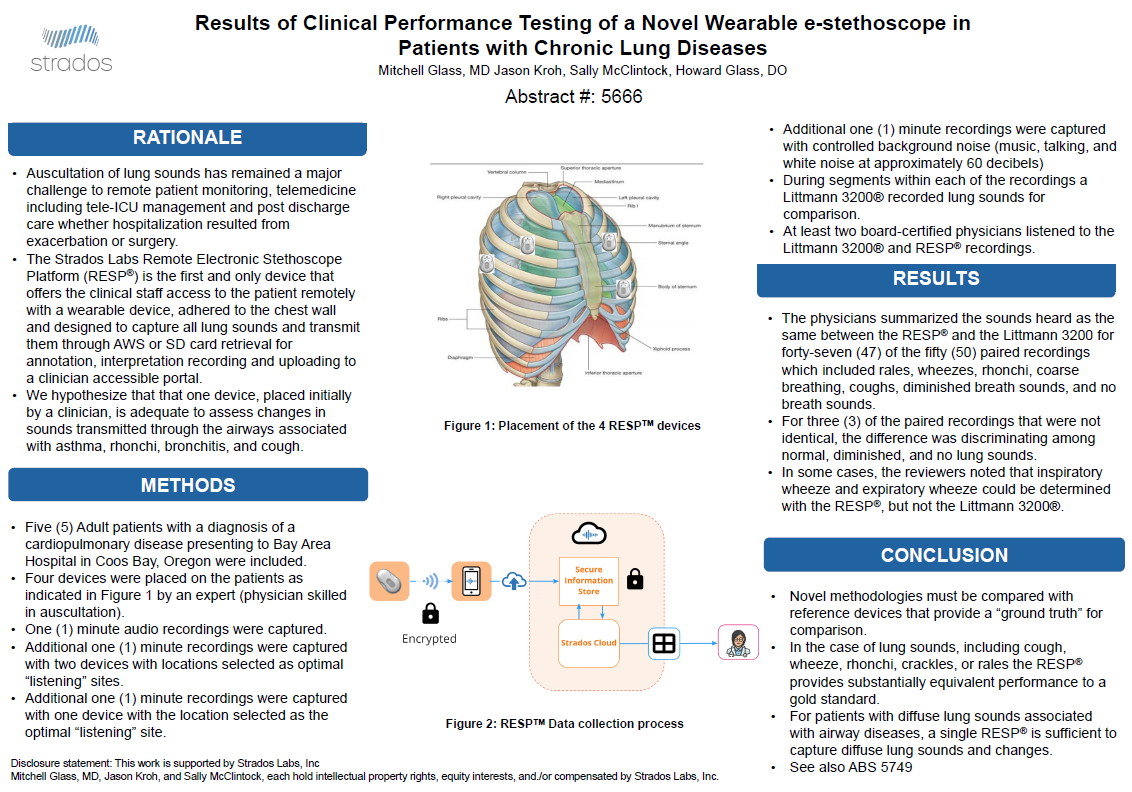
- The gold standard for diagnosing COPD and asthma (auscultation with a stethoscope) is limited to in-person visits, prone to interobserver variability and not archivable
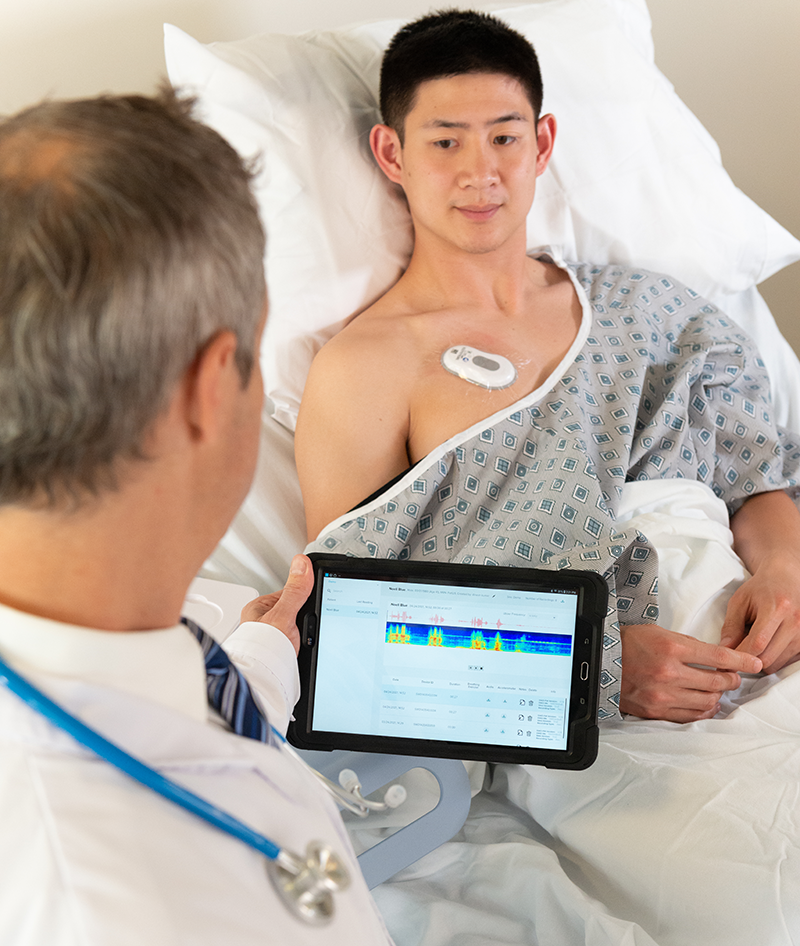
Patient-Centered Design
The RESP Biosensor is small, unobtrusive and hands-free—patients even forget they’re wearing it. Our passive, ‘set and forget’ device captures lung sounds continuously whether patients are sleeping, working out or enjoying time with family. That translates into more patient compliance and more complete data for clinicians and care teams.
Data Reporting
Patient recordings are transferred to the cloud for processing and analysis by our team. Proprietary machine learning algorithms are used to provide varying levels of automation in the annotation process.*
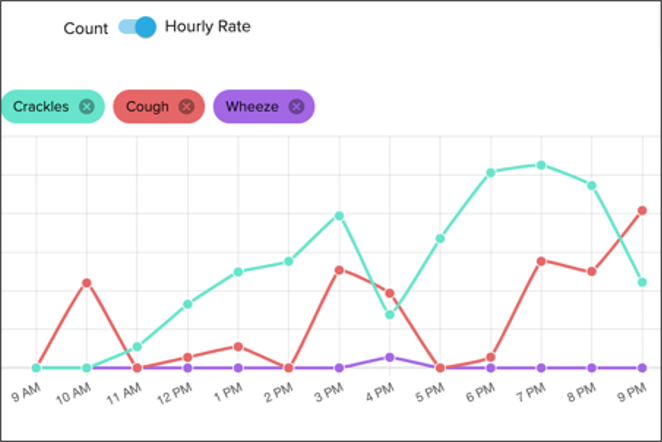
*Machine learning algorithms not cleared by FDA
Clinical Validation
Explore our peer reviewed performance testing, studies and publications.
View more of our research
Let’s Talk.
We’d love to know how we can help you and your patients.