Cough Monitoring
Strados provides leading solutions for assessing cough burden in research and clinical trials, delivering objective, comprehensive insights while reducing site and patient burden.

RESP® Biosensor
FDA 510(k) Cleared
High-fidelity, multi-dimensional cough monitoring biosensor.

Watch-Based Monitoring
RESP® Watch
Low-burden, wrist-worn cough monitoring for multi-day studies.
Capabilities
Incorporates end-to-end encryption and speech obfuscation to preserve patient privacy.
Cloud-based data syncs shorten time to cough analysis before devices are returned.
FDA 510(k) class II medical device designed to capture subject coughs only (RESP Biosensor).
Wireless, discreet device designed to capture authentic symptom burden in daily life.
Goes beyond cough count to provide a complete view of subject cough burden.
Choose between the RESP® Biosensor and Watch-based monitoring for the right fit for each study design.
Product Family
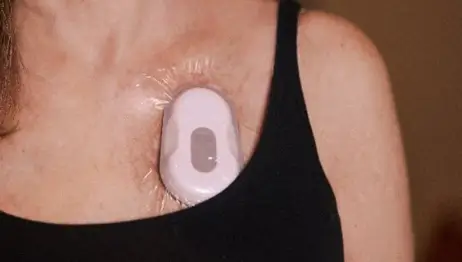
FDA 510(k) Cleared · CE Marked
FDA-cleared medical device designed for high-fidelity, multi-dimensional cough monitoring.
Learn more
Clinical Trial Ready
Wrist-worn device designed for low burden, multi-day cough monitoring.
Learn more*Measurement not FDA cleared but may be used in clinical trials.
Intelligence Advantage
Strados Labs' trained and qualified annotation team identifies and counts cough and lung sound events from subject recordings to meet primary endpoint requirements. Our CoughCheck™ machine learning algorithm, trained on over 200,000 coughs, is available for high-volume screening or exploratory endpoints.
Learn about the Intelligence AdvantageRetained audio
Full audio evidence chain preserved from capture through to audit.
AI-driven event detection
CoughCheck™ algorithm trained on 200,000+ coughs for high-volume screening.
Hybrid annotation
Expert annotation alongside AI — not AI replacing experts.
Publications
View our clinical validation, whitepapers and case studies

Cough Detection Performance of the CoughCheck Algorithm in Subjects with Subacute Cough
Poster Presentation, American Thoracic Society, 2025
Objective Cough Monitoring Detects More Patients with Cough Than Patient Self-Reporting
Poster Presentation, American Cough Conference, 2023
Results of Clinical Performance Testing in Chronic Lung Diseases
Poster Presentation, American Thoracic Society, 2022
The Strados Remote Electronic Stethoscope Platform: a novel wearable remote auscultation system
Poster Presentation, American Thoracic Society, 2020
Tell us about your program and we'll help you design the right cough endpoint strategy.
Design a Study →