Strados Labs RESP® Biosensor
The RESP® Biosensor enables life sciences teams to objectively measure respiratory symptoms and physiological signals in daily life, providing greater insight into treatment response and differentiation across clinical programs.
Design a Study
RESP® Biosensor Inspiration
As we developed a solution to improve the surveillance of chronic respiratory diseases, we designed the RESP® Biosensor to continuously capture lung sounds, extending the reach of traditional stethoscope auscultation and providing clinical and research teams with objective insights from anytime, anywhere.

Traditional Auscultation
Traditional Auscultation
Clinician-dependent, subjective assessment
No continuous overnight monitoring
Limited to scheduled appointment windows
No remote data capture
RESP® Biosensor
Continuous capture anytime, anywhere
Objective, algorithmic event detection
24/7 monitoring including sleep
Real-world, daily-life data
Automated cloud data transfer
RESP® Biosensor
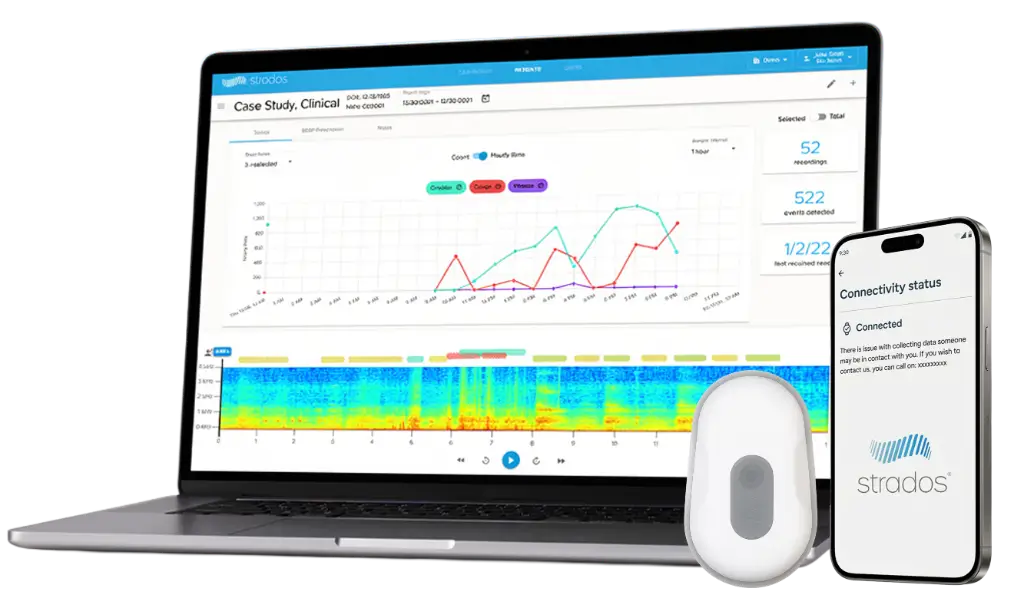
The RESP® Biosensor, often described as the 'Holter monitor for the lungs', is designed to continuously capture high-fidelity cough, lung sounds, and physiological measures to provide greater insight into respiratory status from anywhere, anytime. Clinically validated with equivalent performance to standard electronic stethoscopes.¹
Wireless, discreet form factor and privacy-preserving — designed for comfortable multi-day wear without disrupting daily life.
Comprehensive respiratory and physiological measures that matter to patients, captured passively across the full monitoring period.
Automated or manual (human) overread to meet endpoint requirements. Comprehensive reports prepared by the Strados Labs team.
¹ M. Glass, et al. Results of Clinical Performance Testing of a Novel Wearable e-Stethoscope in Patients with Chronic Lung Diseases [abstract]. Am J Respir Crit Care Med
Strados Intelligence Advantage
The RESP® Biosensor combines advanced acoustic monitoring with an integrated inertial measurement unit to capture lung sounds alongside chest wall movement, respiratory rate, and activity levels. Every event is processed through our proprietary CoughCheck™ algorithm and reviewed by trained human analysts — combining the scale of automation with the precision of expert oversight to produce regulatory-grade endpoint data.
RESP® Biosensor
RESP® Biosensor Measures
A robust combination of measures that reflect how patients feel and function. Onboard inertial measurement unit (IMU) allows for physiological insights alongside cough and lung sounds.
Cough Frequency (24-hour)
Cough Bouts
Cough Sound Intensity
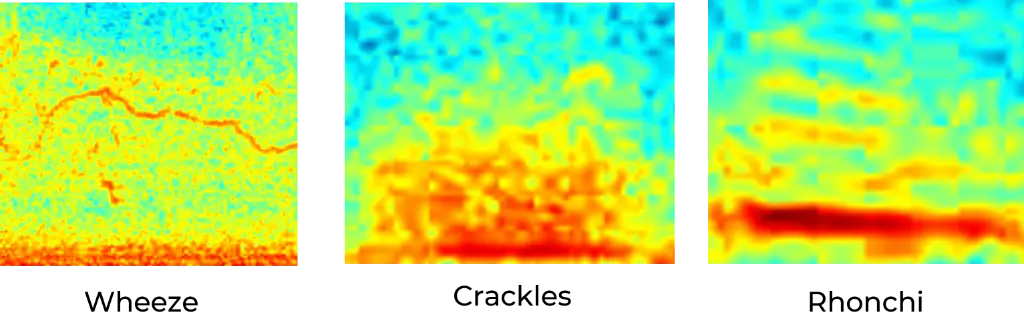
Wheeze, Rhonchi, Crackles
Breathing Patterns*
Respiratory Rate*
Heart Rate & Heart Rate Variability*
Orientation (e.g. Supine or Upright)*
Sleep/Wake*
Activity Levels*
*Measurement not FDA cleared but may be collected in clinical trials

RESP® Biosensor
*Measurement not FDA cleared but may be collected in clinical trials
Clinical Rationale
In increasingly crowded respiratory markets where traditional endpoints often converge, continuous cough and lung sound monitoring offers a powerful opportunity to differentiate and gain unprecedented insights into treatment response.
Improve insight into how patients feel and function, strengthening evidence to regulatory agencies
Reduce reliance on questionnaires to demonstrate treatment effect on symptoms
Differentiate your asset in a competitive marketplace
Uncover signals that may point to new or expanded indications
Identify new pharmacologic insights into your treatment
RESP® Biosensor


RESP® Biosensor
View our clinical validation, whitepapers and case studies
View all publications
Cough Detection Performance of the CoughCheck Algorithm in Subjects with Subacute Cough
Poster Presentation, American Thoracic Society, 2025
Objective Cough Monitoring Detects More Patients with Cough Than Patient Self-Reporting
Poster Presentation, American Cough Conference, 2023
Results of Clinical Performance Testing in Chronic Lung Diseases
Poster Presentation, American Thoracic Society, 2022
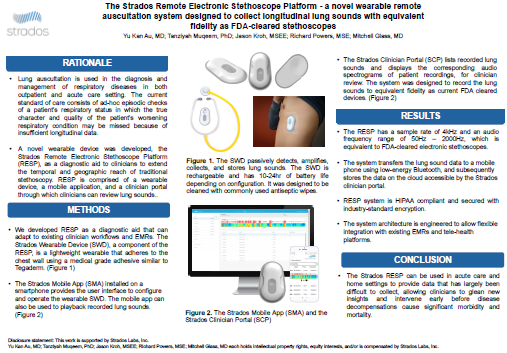
The Strados Remote Electronic Stethoscope Platform: a novel wearable remote auscultation system
Poster Presentation, American Thoracic Society, 2020
Endpoint Suite
In-clinic and at-home pulmonary function testing.
Fractional exhaled nitric oxide for airway inflammation assessment.
Continuous, objective cough monitoring with wearable sensors.
Electronic clinical and patient-reported outcomes.
Speak with our team to learn how objective cough and lung sound monitoring can strengthen your clinical trial endpoints.
Talk to Our Team