Supported by the Air Next Spirometer
FDA 510(k) Cleared | CE Mark
Strados supports both investigator-site primary endpoints and continuous at-home monitoring — same device, same data quality, one integrated workflow.
Design a Study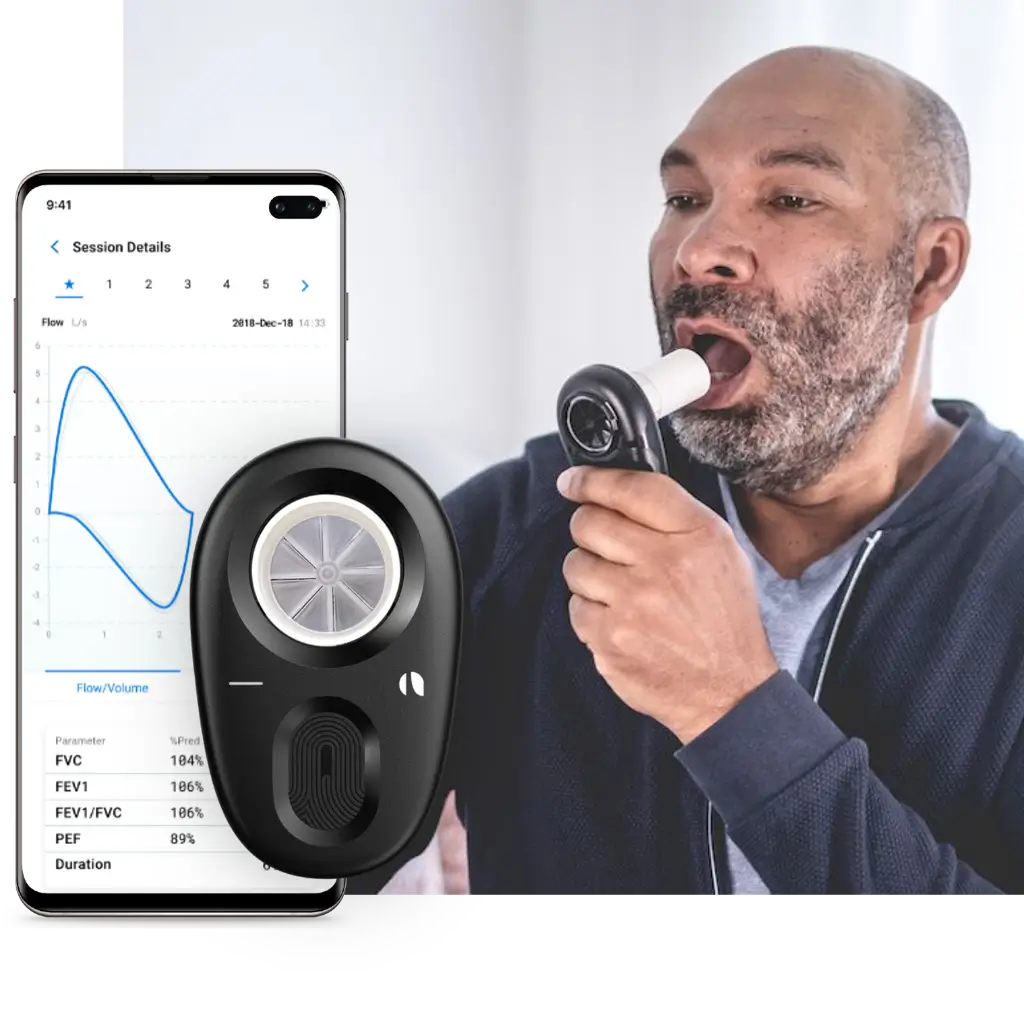
ATS/ERS 2005 & 2019 Compliant
All spirometry meets the latest international acceptability and repeatability standards
Expert Overread
Centralized quality review by trained spirometry specialists for every test
In-Clinic & At-Home
Same device supports primary endpoints at site and continuous at-home monitoring
Remote Telehealth Coaching
Real-time video coaching and app-guided instructions for at-home maneuvers
Deployment Modes
The Strados Air Next supports both investigator-site primary endpoints and continuous at-home monitoring — same device, same data quality, one integrated workflow.
Investigator-site spirometry that supports primary and key secondary endpoints in pivotal trials. Technician training, calibration and quality control, and centralized expert overread ensure data meets the highest regulatory standards.
Extend pulmonary function testing into daily life between clinic visits. The Strados remote telehealth platform provides real-time video coaching and app-guided instructions so patients perform maneuvers correctly — from home.
Measures
Comprehensive pulmonary function parameters meeting ATS/ERS 2005 & 2019 acceptability and repeatability standards.
Available Parameters Include:
FEV₁
Key endpointForced Expiratory Volume in 1 second
FVC
Key endpointForced Vital Capacity
FEV₁/FVC
Key endpointTiffeneau-Pinelli index
PEF
Key endpointPeak Expiratory Flow
FEF25-75%
Forced Expiratory Flow at 25–75% of FVC
All measures meet ATS/ERS 2005 & 2019 acceptability and repeatability criteria.
Standards Compliance
Acceptability & repeatability criteria for FVC maneuver
Updated quality grading and back-extrapolation standards
Electronic records & audit trail compliance
Patient data privacy and security
Full-Service Spirometry
Strados delivers a complete spirometry service — from hardware to human expertise.
The Strados Air Next with companion smartphone app for guided maneuver instructions and real-time patient feedback.
Centralized review by trained spirometry specialists. Every test is assessed for acceptability, repeatability, and quality grading.
End-to-end calibration management and QC protocols at every investigator site to maintain data integrity throughout the trial.
Comprehensive training programs for site staff, covering ATS/ERS maneuver technique, device operation, and troubleshooting.
Full validation documentation, IQ/OQ/PQ, and audit-trail support to meet regulatory submission requirements globally.
Real-time video coaching for at-home spirometry sessions, ensuring patients perform maneuvers correctly without clinic visits.
Dedicated support throughout the trial lifecycle — from site activation to patient onboarding and ongoing helpdesk access.
End-to-end device management including shipping, inventory tracking, returns, and replacements across all global trial sites.
Strados spirometry credentials span 55+ clinical trials, 150+ global sites, and 45+ peer-reviewed publications. Strados spirometry capabilities are powered by our 2025 acquisition of NuvoAir's clinical trial business.
55+
Clinical Trials
234+
Global Sites
34+
Countries
45+
Publications
The Air Next eliminates the need for fixed workstations — the same FDA-cleared device supports ATS/ERS-validated spirometry at investigator sites, in patients' homes, and anywhere in between.
Virtual patient and visit management with fully integrated video and messaging services, replicating the in-person coaching experience for at-home maneuvers.
Spirometry data connects wirelessly and is securely transferred to the cloud in real-time, enabling immediate quality review, risk-based monitoring, and rapid decision-making.
Beyond spirometry, the platform also supports FeNO, cough monitoring, ePRO/eCOA, pMDI inhaler sensors, and SpO₂ — all within a single unified clinical trial environment.
Strados Air Next: FDA-cleared · CE marked · ISO 13485 certified · Meets ATS/ERS/ISO waveform testing requirements
Strados Intelligence Advantage
The Air Next eliminates the need for fixed workstations — the same FDA-cleared device supports ATS/ERS-validated spirometry at investigator sites, in patients' homes, and anywhere in between.
Endpoint Suite
Continuous cough frequency and lung sound monitoring via the RESP® Biosensor and watch-based options.
Fractional exhaled nitric oxide for airway inflammation assessment.
Electronic clinical and patient-reported outcomes.
Advanced acoustic analysis of lung sounds for respiratory assessment.
Speak with our team to learn how Strados Spirometry can strengthen your pulmonary function endpoints.
Design a Study