Strados FeNO
NObreath® — Point-of-Care FeNO Measurement
A rapid, reliable biomarker for Type 2 airway inflammation. FeNO measurement provides objective evidence of eosinophilic inflammation to support patient stratification, endpoint assessment, and treatment monitoring in respiratory trials.
Design a Study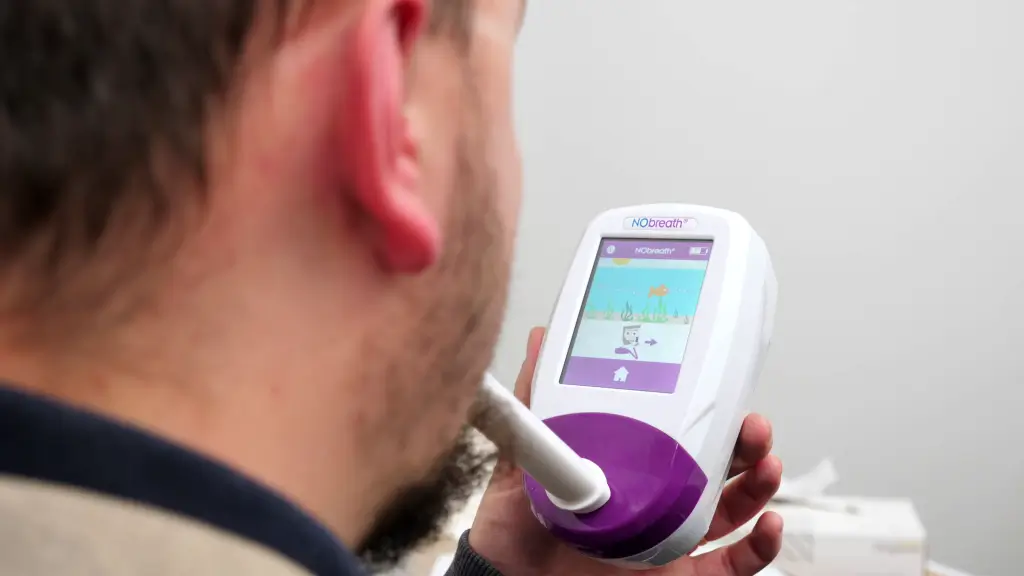
Capabilities
Results available within seconds at the point of care. No lab processing or sample shipping required.
Identify and enrich for Type 2 high patients at screening to improve trial population selection.
Track inflammatory response over time to demonstrate pharmacodynamic effect of anti-inflammatory therapies.
Established correlation with eosinophilic airway inflammation. Recognized by ATS/ERS clinical guidelines.
Non-invasive breath test with guided flow rate control for reproducible measurements across sites.
Compact, portable design for easy deployment across global trial sites with minimal site training.
Clinical Value
FeNO provides a non-invasive, reproducible measure of airway inflammation that complements pulmonary function and patient-reported outcomes.
1
Use FeNO at screening to select patients with Type 2 inflammation, improving response rates and signal detection.
2
FeNO change from baseline provides objective evidence of anti-inflammatory mechanism of action.
3
FeNO captures inflammatory status that spirometry alone cannot assess, adding dimension to your endpoint strategy.
4
Better patient selection through FeNO-based enrichment can reduce the number of subjects needed for adequate power.
Strados Intelligence Advantage
NObreath® offers consistent global availability, portable design, and minimal site training — solving the supply constraints that often limit Niox deployment in clinical trials. Strados partnerships ensure dedicated supply chains and white-glove logistics support for uninterrupted enrollment.
Part of the Strados Endpoint Suite
Combine FeNO with spirometry, cough monitoring, and eCOA to build the most complete picture of respiratory disease status and treatment response.
Tell us about your program and we'll help you design the right respiratory endpoint strategy.
Design a Study