FOR RESEARCH & CLINICAL TRIALS
RESP® Biosensor
Digital respiratory endpoints and cough monitoring for life sciences
FDA 510(k) Class II | CE Mark | HIPAA Compliant
Technology Overview
The RESP® Biosensor is a first-of-its-kind respiratory wearable developed to capture novel insights into treatment effects, including changes in cough frequency and lung sounds such as wheeze.
- Patient-Friendly: A wireless, discreet form factor that respects patient comfort in daily life
- Multi-Parameter: Collects a robust combination of respiratory and physiological measurements
- Validated Reporting: Our qualified and trained annotation team ensures reporting meets endpoint requirements
RESP® Biosensor Measures

-
-
-
-
- Cough Frequency (24-hour)
- Cough Intensity* & Bouts
- All Lung Sounds
- Wheeze
- Rhonchi
- Crackles
- Breathing Patterns & Respiratory Rate*
- Heart Rate & Heart Rate Variability*
- Orientation*
- Sleep/Wake*
- Activity Levels*
-
-
-
*Measurement not FDA cleared but may be collected in clinical trials
Metrics-at-a-Glance
1M+
Lung Sounds Captured
30M+
Breaths Captured
32
Countries
35
2
1
Why the RESP® Biosensor System?
Privacy & Security
Incorporates end-to-end encryption and speech obfuscation to preserve patient privacy
Accelerated Data Turnaround
Cloud-based, remote syncs streamline subject reporting and enable real-time adherence insights
High Fidelity & Integrity
Clinically validated, FDA 510(k) class II medical device that accurately collects data in real-world environments
Patient-Friendly
Wireless, discreet biosensor designed to integrate seamlessly into daily life
Multi-Dimensional Data
Collects a robust combination of respiratory and physiological data, all in one device
More Than a Cough Monitor
Using a combination of audio and motion data, the RESP Biosensor unlocks valuable insight across multiple indications, offering a more complete picture of subjects’ respiratory status and health-related quality of life alongside PROs.
| Cough | Wheeze | Crackles | Respiratory Rate* | Heart Rate* | Activity Level* | Orientation* | |
| Chronic Cough | |||||||
| IPF | |||||||
| Bronchitis | |||||||
| COPD | |||||||
| Asthma | |||||||
| RSV | |||||||
| Covid-19 | |||||||
| Heart Failure |
- Chronic Cough
- IPF
- Bronchitis
- COPD
- Asthma
- RSV
- Covid-19
- Heart Failure
*Measurement not FDA cleared but may be collected in clinical trials
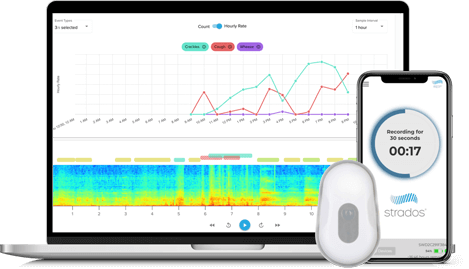
Flexible Reporting—Automated or Human Annotation
- Strados Labs’ trained and qualified annotation team identifies and counts cough and lung sounds from subject recordings to meet primary endpoint requirements.
- Our CoughCheck™ machine learning algorithm*, trained on over 200,000 coughs, has demonstrated strong accuracy in cough detection and is available for applications such as high-volume screening or exploratory endpoints.
*Algorithm not FDA-cleared but may be used in clinical trials
Visual spectrograms of lung sound events facilitate overread for trained annotators
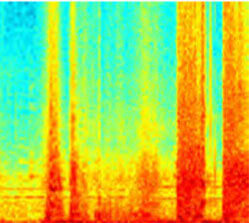
Cough
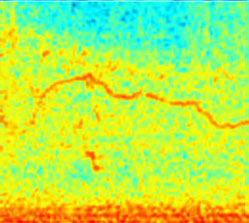
Wheeze
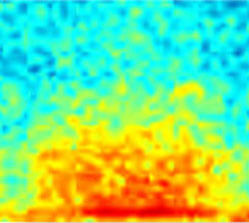
Crackles
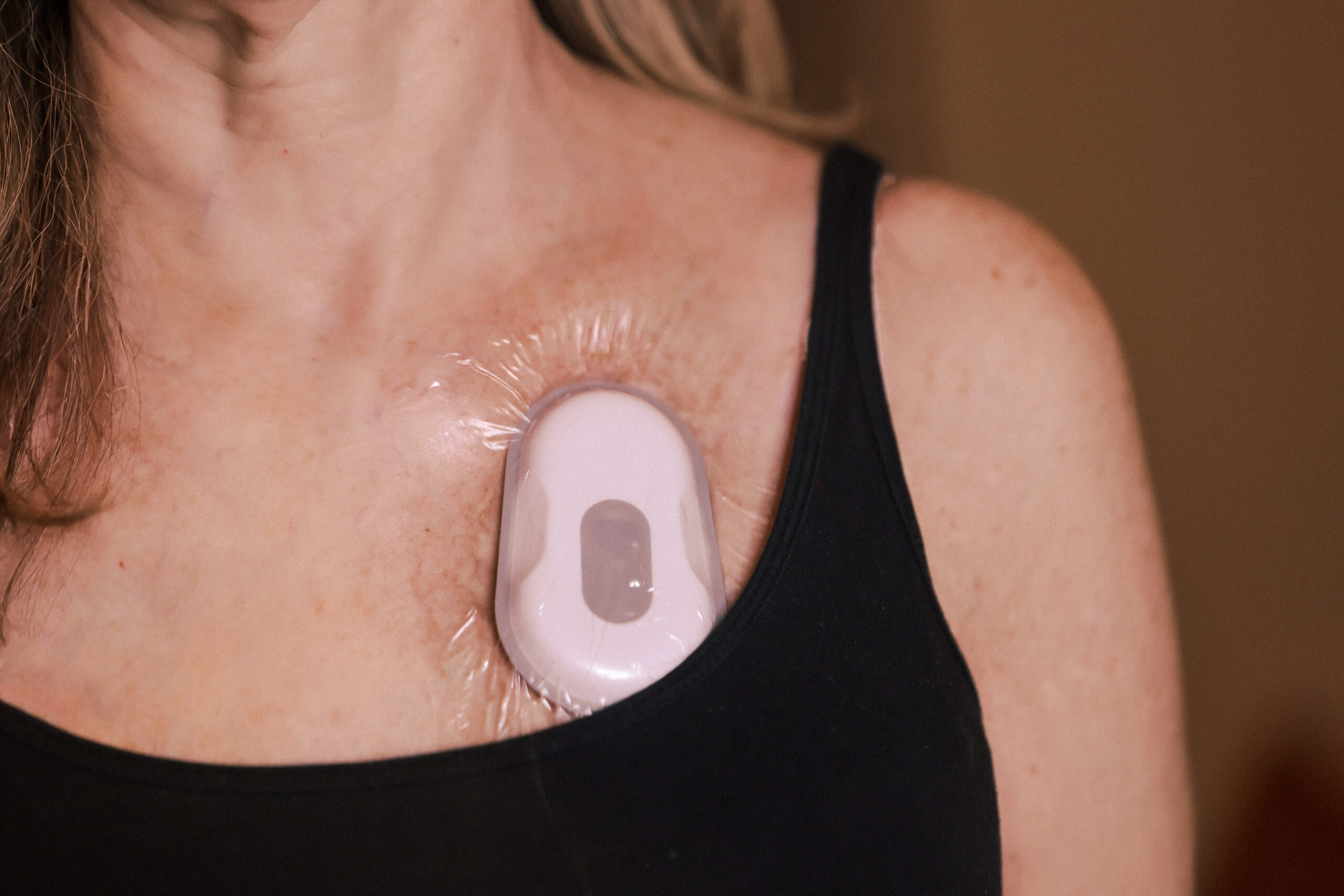
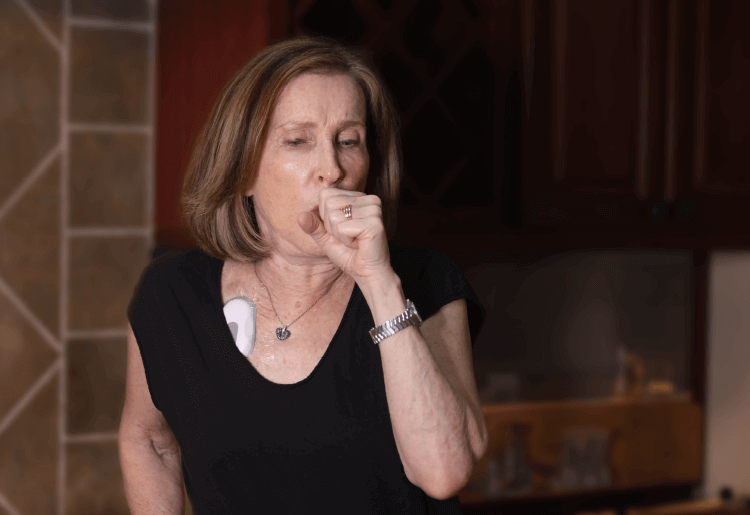
Patient-Friendly Form Factor
- Unobtrusive, wireless form factor reduces patient burden in clinical trials.
- 3M Tegaderm dressing, gentle and breathable, is used to adhere biosensor to chest
- Discreet design helps ensure patients behave naturally, capturing authentic symptom data that mirrors real-life conditions and minimizes the Hawthorne effect.
Publications
View our clinical validation, whitepapers and case studies
Cough Detection Performance of the CoughCheck Algorithm in Subjects with Subacute Cough
Poster Presentation, American Thoracic Society, 2025
Poster Presentation, American Cough Conference, 2023
Talk to our Team
Explore using the RESP® Biosensor in your next study