IPF is chronic lung disease characterized by progressive and irreversible scarring of the lung interstitial tissue. As the scarring worsens it leads to deterioration in pulmonary function and eventually respiratory failure. Historically, IPF trials and treatments have focused on pulmonary function, however, regulators are increasingly asking sponsors to focus on the symptoms that matter most to patients.
For IPF patients, cough is among the most significant and impactful symptoms
Most people have experienced cough at some point, but for patients with IPF, it takes over their lives and is far more than just an annoyance. This is true of many respiratory diseases, but in IPF, cough is particularly prevalent, with over 90% of patients reporting it as a symptom.1 The significant impact of IPF-related cough on patients’ quality of life was highlighted in the FDA’s “The Voice of the Patient” report. Of the 60 patients involved, more than 75% described cough as one of the symptoms that mattered most, alongside shortness of breath and fatigue.2
“On my worst days, cough will wipe me out for an entire day… physically I’m exhausted”
-IPF patient
“I can only sleep for maybe an hour and half or two hours before I have to get up and cough”
-IPF patient
Patients not only described the acute impact of coughing bouts, but also how cough impacts nearly all other aspects of their lives, including sleep, exercise and socializing. Given its severe impact, addressing cough represents a meaningful opportunity for clinicians and sponsors to improve patients’ quality of life.
Cough relief remains a significant unmet need in IPF
Despite the prevalence and burden of cough in IPF, it remains an unmet need for patients. Antifibrotic agents, the current standard of care for IPF, are primarily focused on improving lung function and reducing mortality. Registrational trials for these therapies have demonstrated improvements in lung function as measured via FVC and reduced mortality; however, none included any analysis of cough.3-5 Outside of these trials, evidence that antifibrotic treatments may also improve cough is limited and conflicting.6-9
Beyond antifibrotic treatments, cough suppressants are an option; however, there is very little data evaluating these therapies specifically in IPF, and available evidence suggests limited efficacy, tolerability concerns, and potential risk of addiction with long-term use.10,11
What recent studies agree, is that there is a lack of approved treatment options for cough in IPF. Particularly given that cough is one of the symptoms that matters most to patients.9,12
“There is an urgent need to identify more effective therapies to prevent worsening and ultimately improve how patients feel and function”12
NIH, FDA, Patients & Investigators 2023 Symposium on endpoints in IPF trials
“The lack of specific treatments and management for cough in these patients remains a significant unmet clinical and patient need”9
The burden of cough in IPF and other interstitial lung diseases, Green et al., 2024
How can we measure cough in IPF?
Cough in IPF trials can be measured using subjective or objective measures. Expert consensus recommends the combined use of both, as each provides complementary information and helps build a reliable and clinically meaningful picture.8,13,14
Questionnaires Used in IPF Studies
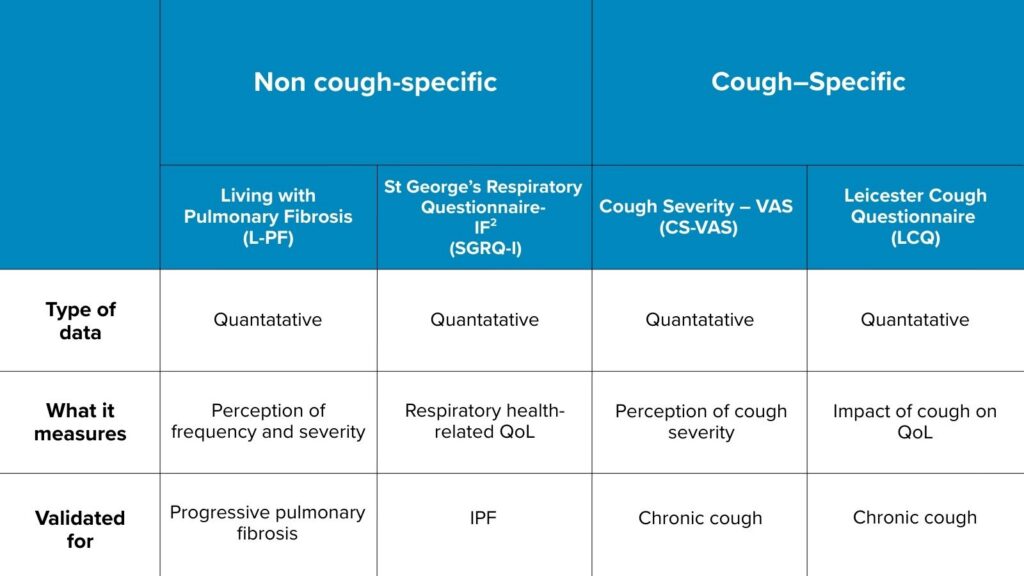
The most commonly used patient reported outcomes (PROs) in IPF provide crucial insight into cough frequency and severity, but especially quality of life. However, as with all subjective measures, their reliability depends on the patient’s recall, interpretation and potential reporting bias.
Due to some of the inherent limitations of PROs, objective cough monitoring is also recommended to provide the most complete picture of patient cough burden. Objective cough monitors are focused primarily on capturing the total number of cough events over a given period. All devices available collect cough sounds using slightly different approaches. The RESP® Biosensor uses a stethoscope-type sensor combined with an accelerometer, allowing it to capture all lung sounds including cough.
Additional measures of cough such as cough bouts and sound intensity have also been explored to provide greater insight into cough burden. Patients and clinicians alike recognize that defining cough burden solely by frequency is limited.15 Two patients with the same cough frequency may experience vastly different levels of severity, quality of life impact, and clinical risk depending on the patterns and intensity of their cough.
Recent studies using the RESP® biosensor have demonstrated that cough intensity is higher during cough bouts, and greater in chronic vs. subacute cough, suggesting that intensity may offer a valuable marker of disease severity and burden (deLaubenfels et al, 2025, deLaubenfels et al, 2025).
Designing future trials to address the unmet needs in IPF
Patients, clinicians, regulators and the literature agree that more data and better treatment options for treating cough in IPF are urgently needed.2,8,9,12 As sponsors react to meet this need, incorporating both subjective and objective cough measures will be essential to generate robust, clinically meaningful evidence. Advances in objective cough monitoring have improved data reliability and patient convenience, while also enabling novel endpoints – such as cough intensity – that may provide deeper insight into cough relief.
References
- Hirons, B. et al. (2023). Cough in chronic lung disease: a state of the art review. Journal of thoracic disease, 15(10), 5823-5843. https://doi.org/10.21037/jtd-22-1776
- U.S. Food and Drug Administration (FDA) (2015). Voice of the patient – Idiopathic Fibrosis.https://www.fda.gov/files/about%20fda/published/The-Voice-of-the-Patient–ldiopathic-Pulmonary-Fibrosis.pdf
- Richeldi L, du Bois RM, Raghu G, et al. Efficacy and safety of nintedanib in idiopathic pulmonary fibrosis. N Engl J Med. 2014;370(22):2071-2082. doi:10.1056/NEJMoa1402584
- King TE Jr, Bradford WZ, Castro-Bernardini S, et al. A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N Engl J Med. 2014;370(22):2083-2092. doi:10.1056/NEJMoa1402582
- Richeldi L, Azuma A, Cottin V, et al. Nerandomilast in Patients with Idiopathic Pulmonary Fibrosis. N Engl J Med. 2025;392(22):2193-2202. doi:10.1056/NEJMoa2414108
- van Manen MJG, Birring SS, Vancheri C, et al. Effect of pirfenidone on cough in patients with idiopathic pulmonary fibrosis. Eur Respir J. 2017;50(4):1701157. Published 2017 Oct 19. doi:10.1183/13993003.01157-2017
- Wijsenbeek M, Swigris JJ, Inoue Y, et al. Effects of nintedanib on symptoms in patients with progressive pulmonary fibrosis. Eur Respir J. 2024;63(2):2300752. Published 2024 Feb 1. doi:10.1183/13993003.00752-2023
- Wu Z, Smith DJF, Yazbeck L, et al. Cough Severity Visual Analog Scale Assesses Cough Burden and Predicts Survival in Idiopathic Pulmonary Fibrosis. Am J Respir Crit Care Med. 2024;209(9):1165-1167. doi:10.1164/rccm.202311-2169LE
- Green R, Baldwin M, Pooley N, et al. The burden of cough in idiopathic pulmonary fibrosis and other interstitial lung diseases: a systematic evidence synthesis. Respir Res. 2024;25(1):325. Published 2024 Aug 27. doi:10.1186/s12931-024-02897-w
- Birring SS, Kavanagh JE, Irwin RS, et al. Treatment of Interstitial Lung Disease Associated Cough: CHEST Guideline and Expert Panel Report. Chest. 2018;154(4):904-917. doi:10.1016/j.chest.2018.06.038
- Vigeland CL, Hughes AH, Horton MR. Etiology and treatment of cough in idiopathic pulmonary fibrosis. Respir Med. 2017;123:98-104. doi:10.1016/j.rmed.2016.12.016
- Raghu et al. (2024). Meaningful Endpoints for Idiopathic Pulmonary Fibrosis (IPF) Clinical Trials: Emphasis on ‘Feels, Functions, Survives’. American journal of respiratory and critical care medicine. 209. 10.1164/rccm.202312-2213SO
- Mann J, Goh NSL, Holland AE, Khor YH. Cough in Idiopathic Pulmonary Fibrosis. Front Rehabil Sci. 2021;2:751798. Published 2021 Oct 18. doi:10.3389/fresc.2021.751798
- Boulet LP, Coeytaux RR, McCrory DC, et al. Tools for assessing outcomes in studies of chronic cough: CHEST guideline and expert panel report. Chest. 2015;147(3):804-814. doi:10.1378/chest.14-2506
- Turner RD, Birring SS. Measuring cough: what really matters? J Thorac Dis. 2023;15(4):2288-2299. doi:10.21037/jtd-23-230