COUGH MONITORING WITH STRADOS LABS
RESP® Biosensor for Clinical Trials
Capture accurate, objective cough and lung sound metrics, driving stronger evidence in respiratory drug development.
FDA 510(k) Class II Device | CE Mark | HIPAA Compliant
- Respiratory symptoms such as cough, wheeze, and crackles are associated with disease severity and quality of life across many cardiopulmonary and infectious diseases.
- Patient self-reports, while valuable, may not fully capture the objective reality these symptoms present.
- The RESP Biosensor detects the full scope of respiratory symptoms, providing robust data to demonstrate treatment efficacy.
Objective Cough Monitoring in Respiratory Research
Unlock 24-hour clinically accurate and continuous monitoring of cough and lung sounds with the RESP Biosensor.* Optimized for ease of use at home as part of clinical trials, our device offers:
- Enhanced Patient Compliance: A wireless, discreet design that respects patient comfort and lifestyle.
- Rapid, Reliable Data Access: Direct cloud sync facilitates efficient analysis of objective respiratory metrics.
- Superior and Comprehensive Data: Captures detailed cough and lung sound data, offering insights beyond traditional methods.
In comparison to bulkier alternatives, the RESP Biosensor delivers a blend of convenience, speed, and accuracy — ensuring your research is both patient-centric and data-rich.
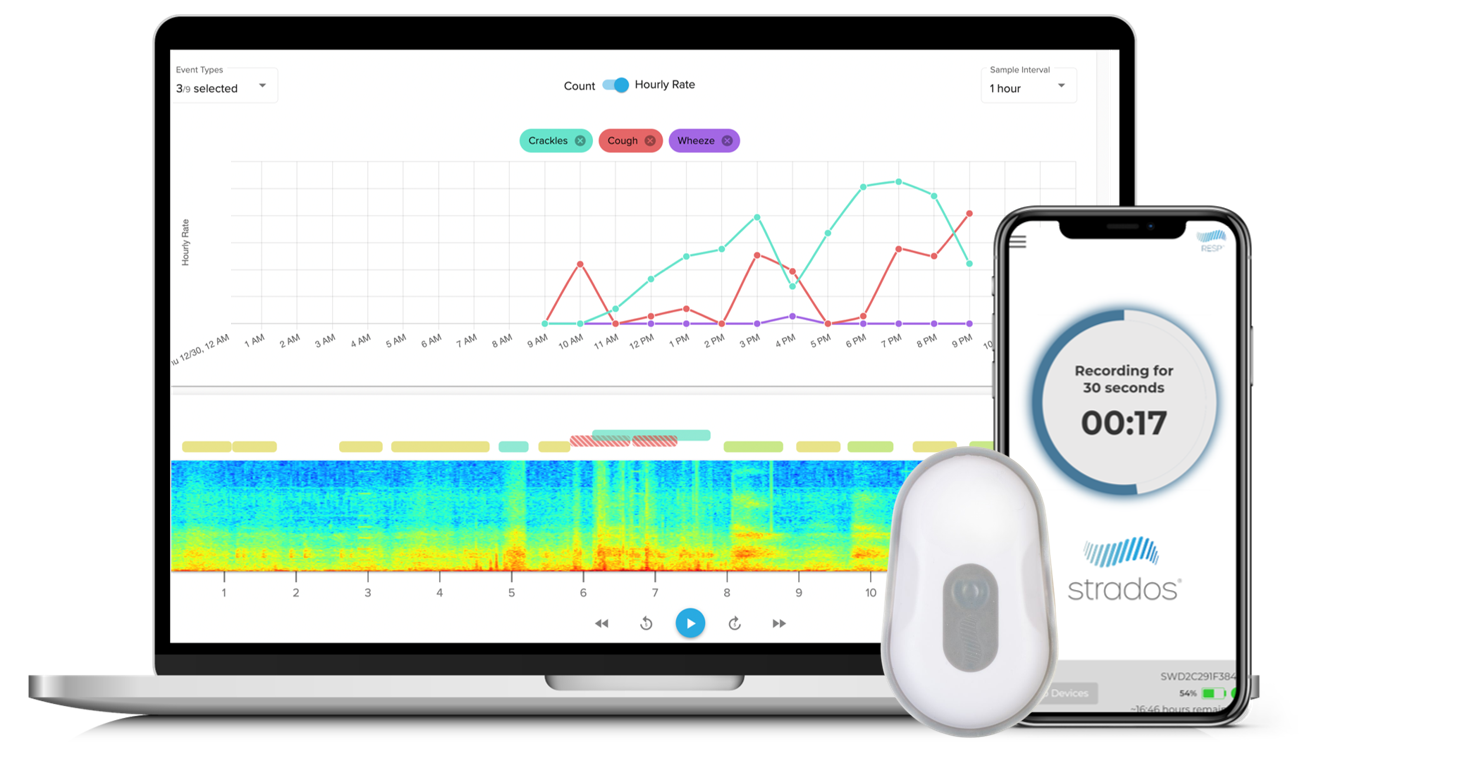
RESP Biosensor Measurements

Cough Monitoring
Accurately assess cough frequency, bouts, and intensity, essential for chronic cough therapies

Lung Sounds
Gain detailed wheeze, rhonchi, and crackles analysis for greater respiratory context.

Additional Insights
Track respiratory rate, activity levels, sleep/wake, and body positioning for quality-of-life metrics.
*Not yet cleared by the FDA
Why Choose the RESP® Biosensor?
Privacy & Security
Incorporates end-to-end encryption and advanced noise filtering for strong patient data privacy.
Accelerated Insights
Real-time, cloud-based syncs reduce the wait time from data collection to actionable insights.
Coughs
The device uses multi-modal data to differentiate between subject coughs and external coughs.
Designed for Daily Life
Low-burden, discreet design aids patient comfort and compliance while mirroring real-life conditions.
Beyond Cough Count:
Extended capabilities beyond cough frequency include intensity, bouts, lung sounds and activity levels for a fuller understanding of patient health.