Strados to Participate in DiMe Project: Building the Business Case for Digital Endpoints

Strados Labs is excited to announce its participation in a groundbreaking project led by the Digital Medicine Society (DiMe), a global non-profit dedicated to driving scientific progress and broad acceptance in digital medicine. The project, Building the Business Case for Digital Endpoints is focused on advancing the use of digital endpoints, such as cough, wheeze and […]
Strados Labs Hits 10 Million Breaths!

We are thrilled to announce that through our various partnerships with life sciences organizations, Strados Labs has successfully captured a staggering 10 million breaths in this year alone – and counting! This milestone marks a significant step towards advancing respiratory care by harnessing the power of data and innovation. Here’s to countless more breaths captured and making […]
Strados Labs Announces Pediatric Asthma Study to Validate its Wearable Device in Children with Wheeze
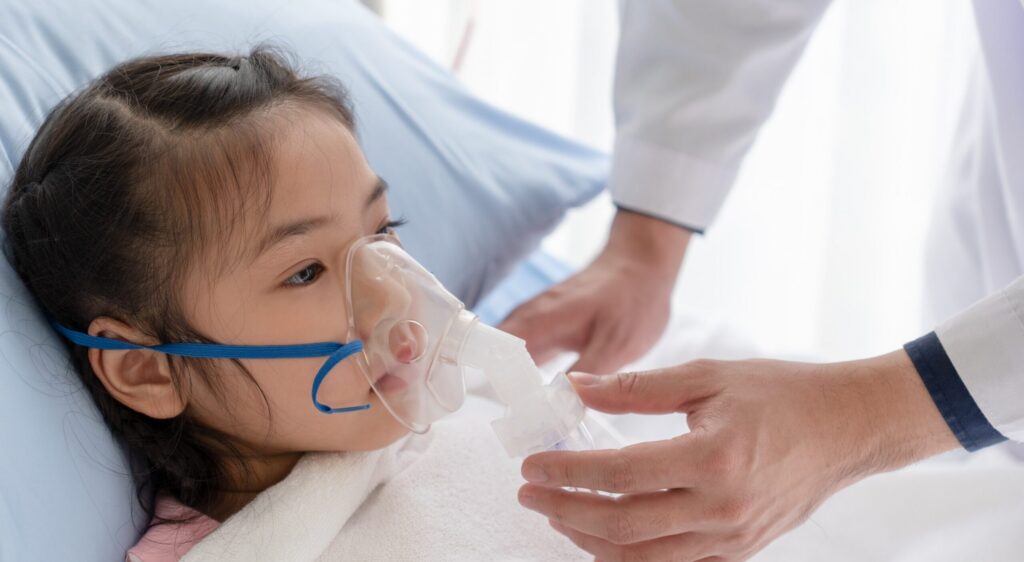
Strados Labs’ RESP™ Biosensor is currently being used in a clinical trial at Lurie Children’s to determine if the technology can accurately detect wheezing and other symptoms of asthma exacerbation in children. PHILADELPHIA, PA – September 26th, 2023 – Strados Labs has partnered with Ann & Robert H. Lurie Children’s Hospital of Chicago for a clinical trial to […]
Strados Labs Recognized as Quarterfinalist for the Digital Health Hub Foundation: Digital Health Awards

PHILADELPHIA, PA – August 31, 2023 – Strados Labs is thrilled to announce its selection as a quarterfinalist for the prestigious 2023 Digital Health Awards, presented by the Digital Health Hub Foundation. Strados Labs was chosen as a quarterfinalist for the Best in Class in the Pediatric-Adolescent Care Innovation Health category. This recognition highlights the company’s dedication […]
Clario and Strados Labs partner to bring new pulmonary endpoints to respiratory clinical trials

Clario has partnered with Strados Labs to offer its customers the RESP™ Biosensor System, an advanced technological innovation for the at-home capture of lung sounds. The revolutionary device enables Clario to support the capture of additional clinical respiratory endpoints such as cough and wheeze, and it does so in a patient-friendly, streamlined way. PHILADELPHIA, PA […]
Strados Wins the 2023 ATA Telehealth Innovators Challenge

Nick Delmonico, Cofounder and CEO of Strados Labs presented live at ATA 2023 on March 6th in San Antonio, TX and was named a winner in the ‘Tools that Deliver Care’ category. Philadelphia, PA March 8, 2023 – Strados Labs today announced that it was named a winner in the ATA Telehealth Innovators Challenge as part of the ‘Tools […]
RESP™ Biosensor Added to Government Contract Vehicles Through Partner, Lovell

PENSACOLA, FLORIDA, UNITED STATES, February 2, 2023 — Strados Labs announced today that their remote monitoring solution, the Strados™ Care Respiratory Platform, which includes the RESP™ Biosensor, can now be found on the Defense Logistics Agency’s Electronic Catalogue for Medical Supply Chain (ECAT), the Department of Veterans Affairs’ Federal Supply Schedule (FSS), and the General Services […]
Strados Labs Forms Collaboration with Boehringer Ingelheim for its Idiopathic Pulmonary Fibrosis (IPF) Pilot Study

Starting in early 2023 the biopharmaceutical company will use the Strados RESP™ Biosensor to remotely monitor cough and crackles in patients at home. Philadelphia, PA January 26, 2023 – Strados Labs today announced a collaboration with Boehringer Ingelheim to remotely monitor patients using its FDA-cleared pulmonary wearable device in an upcoming idiopathic pulmonary fibrosis (IPF) pilot study. In Boehringer […]
Strados Labs Named a Winner of 2022 Best of Sensors Awards

Recognized in the Wearable Category for the first-of-its-kind smart sensor platform for respiratory health, RESP® Philadelphia, PA, July 14, 2022 – Today, Strados Labs announces it has been named a winner in the Wearables category of the 2022 Best of Sensors Awards for its flagship device RESP®. The awards program is presented by Questex’s Sensors Converge and Fierce Electronics and honors the best in […]
Strados Labs Receives FDA 510(k) Clearance for Home Use of its Smart Sensor Platform for Respiratory Health, RESP®

Philadelphia, PA May 3, 2022 — Strados Labs, a medical technology company harnessing the power of smart sensors, machine learning and proprietary algorithms to provide early detection and predictions of worsening respiratory diseases, today announced it has received FDA Class II 510(k) clearance for home use of its first product, RESP®, which make this its second FDA clearance […]
