eCOA
Patient diaries, questionnaires, and clinical assessments
Capture validated patient-reported outcomes, symptom diaries, and quality-of-life instruments digitally — integrated with objective respiratory device data in a single platform.
Design a Study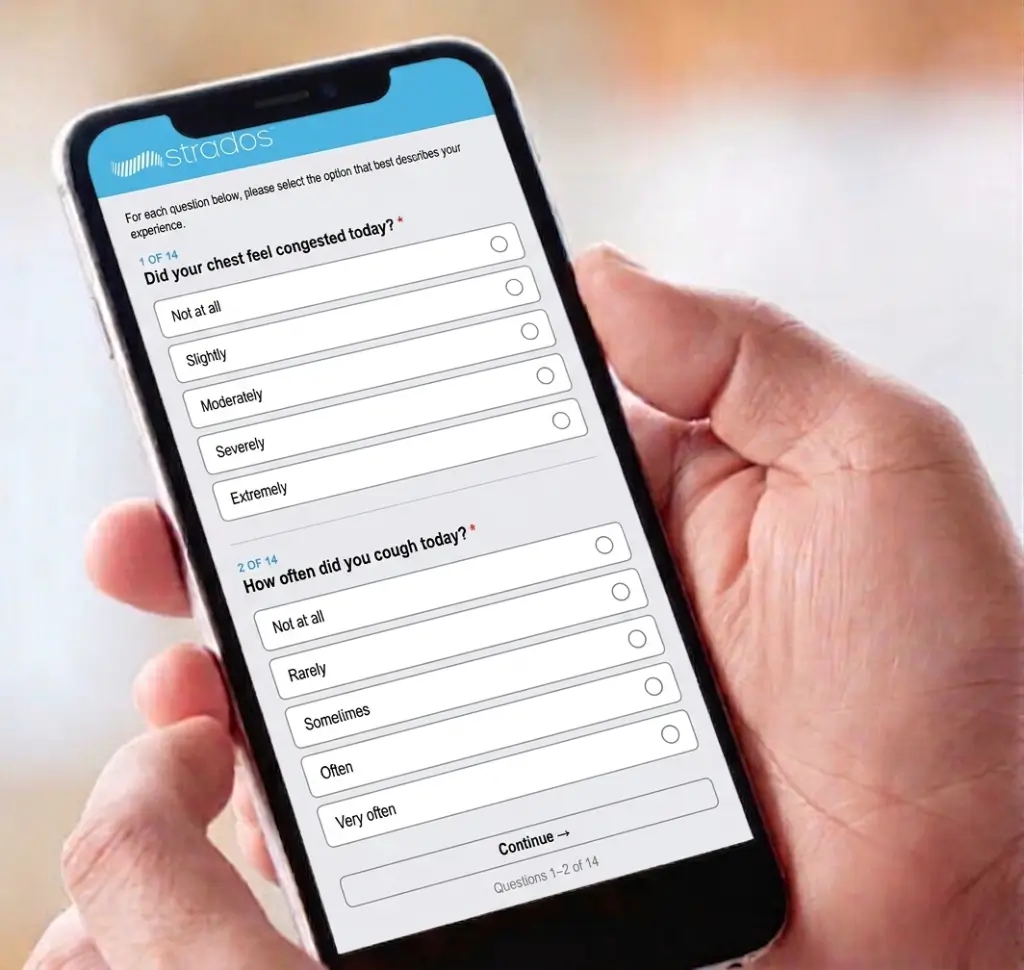
Capabilities
Patients complete assessments on smartphone, tablet, or web. Flexible deployment based on study needs.
Support for standard respiratory PRO instruments including SGRQ, ACQ, AQLQ, CAT, E-RS, and custom questionnaires.
Automated reminders and real-time submission tracking minimize missing data and improve compliance rates.
PRO data flows into the same platform as objective device data, enabling correlated analysis across endpoints.
Audit trails, electronic signatures, and data integrity controls for regulatory compliance.
Daily symptom diaries capture patient experience alongside objective cough, lung sound, and spirometry data.
Clinical Value
Patient-reported outcomes are essential but limited by recall bias and subjectivity. Integrating PROs with objective device data provides a more complete and credible endpoint strategy.
1
Digital diaries with real-time collection minimize the gap between symptom experience and data capture.
2
Correlate patient-reported symptoms with objective cough counts, lung sounds, and spirometry for stronger evidence.
3
Automated reminders, intuitive interfaces, and minimal burden design drive completion rates above 90%.
4
A single data platform for all endpoints means faster analysis, cleaner databases, and more efficient submissions.
Assessment Types
Different perspectives on patient outcomes, all captured digitally and integrated into a single platform.
ePRO
Validated symptom assessments and quality-of-life instruments completed directly by patients, capturing their subjective experience of disease and treatment.
eClinRO
Clinical assessments and observations recorded by healthcare providers using digital forms, standardized scales, and structured data collection.
eObsRO
Structured observations recorded by caregivers or other third parties, capturing their assessment of patient status and treatment response.
eCOA is the layer that connects patient experience with device-measured reality. Together they tell the complete story of treatment effect.
Explore the Full Endpoint Suite