For Bronchiectasis
Gain objective insights into cough burden in bronchiectasis patients
Cough is not just a symptom in bronchiectasis; it is one of the most distressing and persistent clinical features.
-
Up to 90% of patients with bronchiectasis report daily coughing, often with sputum production¹
-
Frequent cough is strongly linked to poorer quality of life, increased airway infection risk, and higher healthcare utilization.²
-
Traditional measures (spirometry, imaging, and patient-reported outcomes) often fail to capture the real-world frequency, severity, and timing of cough.³
For drug developers and clinicians, this gap presents both a clinical challenge and an opportunity: objective cough monitoring can provide granular insights into how treatments impact patient lives.
The RESP® Platform for Cough Monitoring
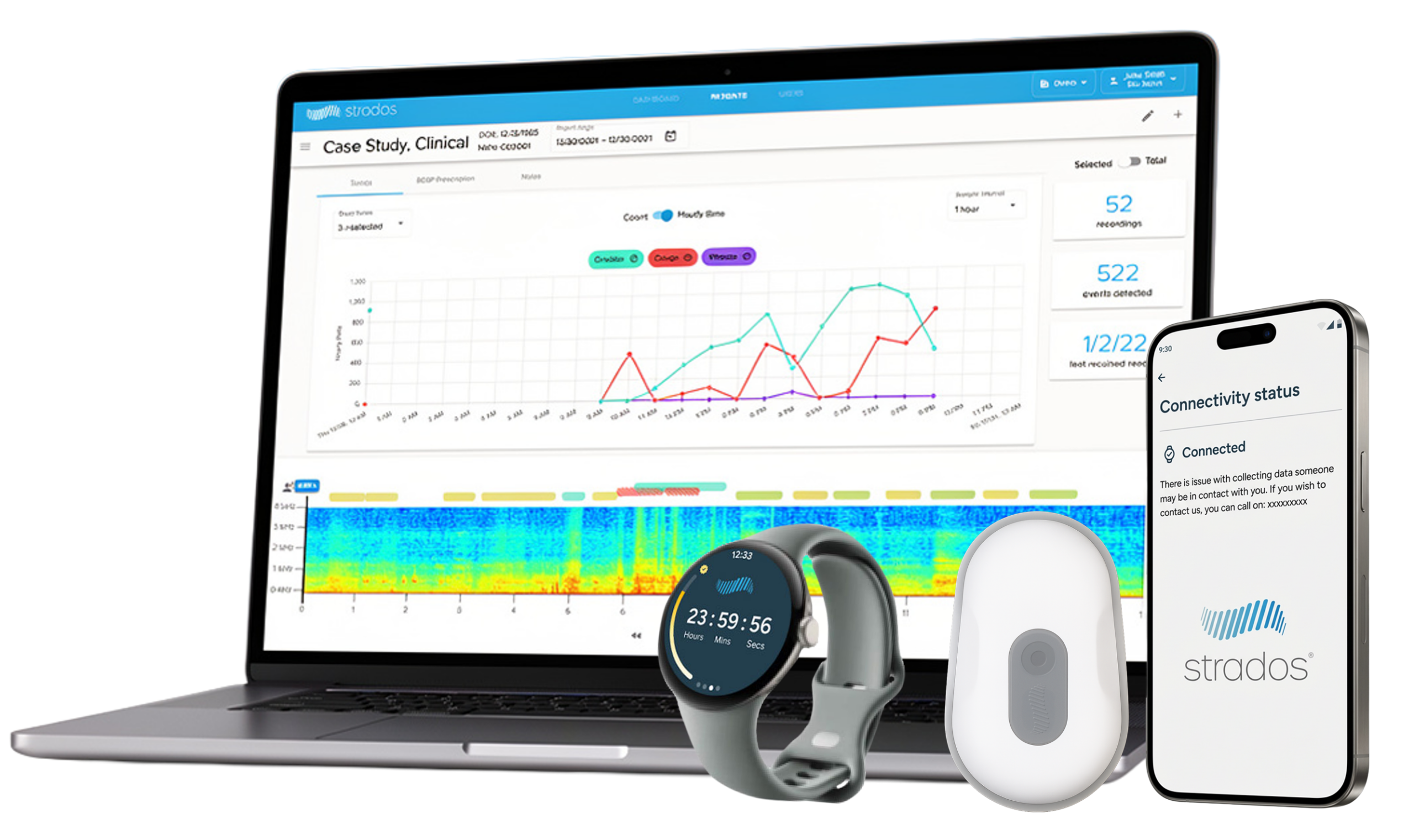
The RESP® Platform is a next-generation, high-fidelity wearable system designed to provide greater insight into patient cough burden in daily life. Our technology offers:
- Flexible device selection for various cough monitoring applications
- Direct cloud syncs that expedite analysis of cough measures and streamline patient reports
- Comprehensive cough insights such as cough frequency, bouts and intensity
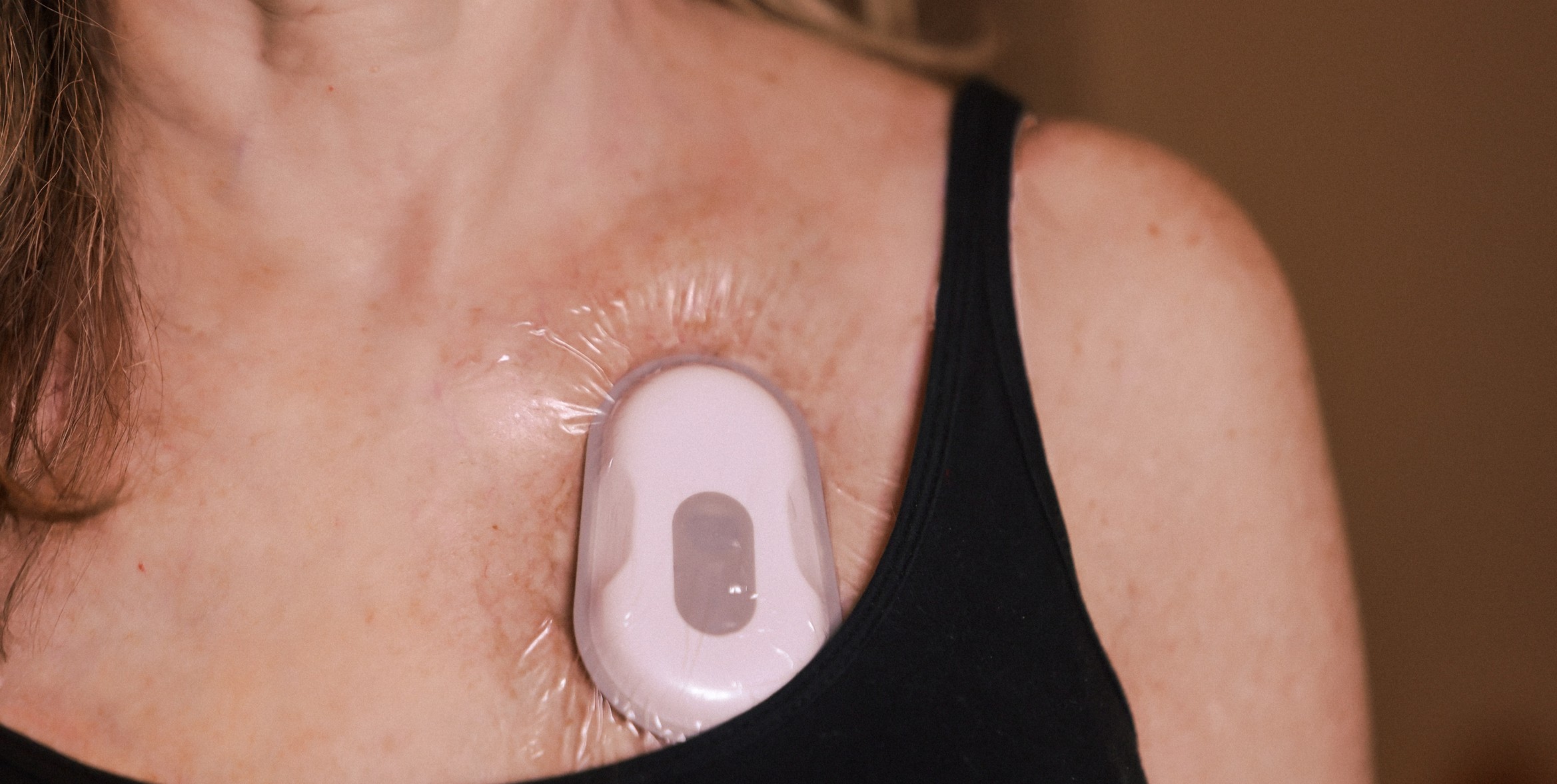
RESP® Biosensor
FDA-cleared medical device designed for high-fidelity, multi-dimensional cough monitoring.
- 24-hour cough frequency
- Cough sound intensity
- Cough bouts
- Sleep/wake*
- Activity levels*
- FDA 510(k) Clearance
- CE-marking
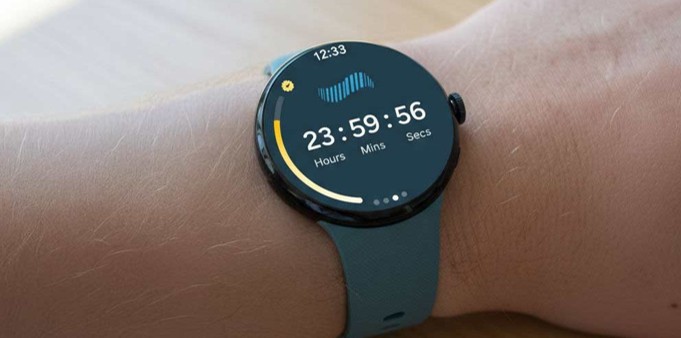
Watch-Based Cough Monitoring
Wrist-worn device designed for low burden, multi-day cough monitoring. For research use only.
- 24-hour cough frequency*
- Cough bouts*
- Heart Rate*
- Respiratory Rate*
- SPO2*
- Skin temperature*
- Sleep Stages*
*Measurement not FDA cleared but may be used in clinical trials
Why the RESP® Platform for Bronchiectasis Patients?
Privacy & Security
Incorporates end-to-end encryption and speech obfuscation to preserve patient privacy
Wireless Data Sync
Cloud-based syncs from paired mobile phones allow for remote data uploads, accelerating subject reporting
High Fidelity & Accuracy
FDA 510(k) class II medical device for capturing lung sounds, including cough
Patient-Friendly
Wireless, discreet biosensor secured by a gentle and breathable adhesive
Advanced Insights
Gain a greater understanding of cough burden beyond cough counts, including cough intensity, breathing patterns, vitals, and other physiological data.
Publications
OVERVIEW VIDEO
“Any time we have the opportunity to diagnose something earlier, it allows us to intervene earlier and potentially change the course of the disease.”
– Albert Rizzo, MD, Chief Medical Officer, American Lung Association
Let’s Connect
Explore using the RESP® Biosensor in your study or practice
References
- Spinou A, Lee KK, et al. The validity of the Leicester Cough Questionnaire in bronchiectasis. Eur Respir J. 2017;49(3):1601532.
- Chalmers JD, et al. The impact of chronic cough in bronchiectasis: relationship to disease severity, quality of life, and exacerbations. Chest. 2014;145(5):974–982.
- McGuinness G, Naidich DP. Bronchiectasis: CT evaluation. AJR Am J Roentgenol. 2002;179(5):1241–1248.
- Guan W, et al. Symptoms predict future risk of exacerbations in adults with bronchiectasis: a multicentre prospective cohort study. Lancet Respir Med. 2025. doi:10.1016/S2213-2600(25)00160-2